What is Virtual Prototyping and Why is it Important in Product Development?
Check out this detailed guide to know about virtual prototyping,...

Product quality doesn’t fail all at once. It slowly breaks down due to small errors, missed checks, and a lack of proper tracking.
In regulated industries, such as food and beverage, medical, and aviation, as organizations grow, it becomes increasingly difficult to maintain consistent product or service quality. Also, relying on scattered notes or memory for a quality check makes it difficult to track what’s working and what’s not.
However, by using the right quality management system, teams can ensure consistent quality for all products being developed. It’s not just for big factories or ISO audits, but small teams can also rely on it to set rules for quality checks.
Whether you are a company owner or from an engineering team that ensures product quality, this guide will help you understand QMS, its key features, how it is used in different industries, and the top QMS of 2025.
A QMS, or Quality Management System, is a software platform that helps businesses track product quality. Whether you’re manufacturing products or providing digital services, QMS helps you to keep your product information organized and easy to follow.
You can think of QMS as a replacement for scattered spreadsheets, official records, and disconnected tools with a single place to manage quality-related work. This is useful in industries like manufacturing, healthcare, aviation, software, and even education.
QMS software is used for:
Did you know?
Deloitte conducted a customer excellence survey in September 2023, and 94% of respondents agreed that it is important to have consistent product quality for customer satisfaction.
Every QMS can have different features according to the industries it serves. However, there are a few core features that every good QMS should contain, which we have covered here:
In highly regulated industries like aerospace, a single oversight can impact aviation performance and safety. By using the QMS software, aviation manufacturers can meet compliance standards like AS9100, ensuring system safety and quality.
Take the example of an aircraft part being built in multiple stages by subcontractors. If any component of the part is not working as expected, the quality assurance team can use the QMS to find the root cause of the issue by using the traceability feature. They can even identify all the parts where failed components were used to stop failures early.
In healthcare, routine procedures are anything but casual. Every process, from laboratory testing to equipment handling, should follow regulatory guidelines. When it is not followed properly, it can significantly impact the patient’s well-being and introduce risk and legal issues.
In this scenario, QMS software should be used by hospitals and device manufacturers to bring some order to a system that otherwise becomes too difficult to manage at scale. It helps in following standards like FDA 21 CFR Part 11, ISO 13485, and GMP.
Furthermore, with QMS, hospitals can manage safety documents and their reviews digitally. Similarly, training completion logs and revision histories are also maintained without relying on memory or informal checks.
#Bonus: If you really want to see how real companies are using QMS in their daily workflow, you can go through this Reddit thread. Here, people are discussing which software and how they’re using it in their company.
To choose the right quality management system, you need to consider the factors below:
After trying multiple QMS, we have picked the 3 best quality management systems given below:
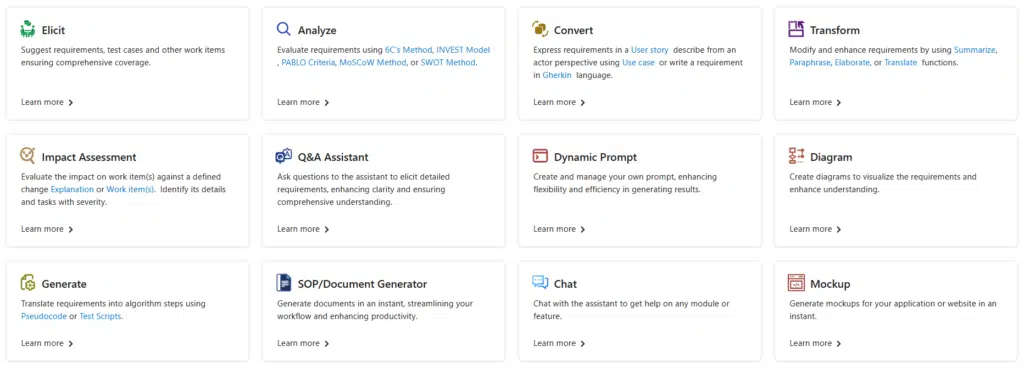
Modern Requirements is designed for teams that work inside Azure DevOps or are planning to shift their quality and requirements workflows into it. It removes the need to juggle multiple tools by combining documentation, compliance, and quality tracking in one place.
Industries It Serves:
Review Rating: 8.8 / 10 (infotech.com)

Ideagen is commonly used in environments where supplier control, incident tracking, and compliance checks need to be handled with care. It fits well in traditional industries where physical products are closely regulated.
Industries It Serves:
Review Rating: 4.3 / 5 (Gartner)
MasterControl is designed for highly regulated production setups, where each action must be recorded, validated, and reviewed. It’s widely adopted in pharma and biotech, especially where electronic batch records and CAPA workflows are involved.
Industries It Serves:
Review Rating: 4.4 / 5 (G2)
Quality management is not a one-time effort, but it is a regular process to maintain the quality of the product. A QMS does not solve everything, but it brings structure to areas that often go overlooked.
Many companies depend on systems, such as spreadsheets, Microsoft Word, etc., that were never built for audits or long-term tracking. Over time, the cracks begin to show. With a good QMS in place, less is left to chance, and more decisions are backed by real data.
Before choosing a platform, look at how your teams actually operate. The best tool is the one that fits without forcing new habits.
If you are using Azure DevOps for project management, try Modern Requirements4DevOps for free and see how it surprises you in quality improvements.
The benefits of QMS are:
The core components of QMS are:
✅ Define, manage, and trace requirements within Azure DevOps
✅ Collaborate seamlessly across regulated teams
✅ Get started for FREE—no credit card required
Check out this detailed guide to know about virtual prototyping,...
Learn more about the importance of SOC 2 compliance, its...
Agents4DevOps puts smart AI agents right into Azure DevOps, letting...