Info-Tech Live 2026
Join Modern Requirements at Info-Tech Live 2026 and witness firsthand...

Medical Software Development is the process through which companies develop apps for both patients and medical professionals. A critical part of success in medical device software development is robust requirements management. These tools help teams balance user needs, business needs, and strict regulations. As the industry becomes more competitive, balancing these needs is getting harder. According to Statista, the medical devices market is projected to grow to $609 billion in revenue by 2028, up from $471 billion today.
As innovation drives the industry forward, the need for precision, compliance, and efficiency is increasing. This article covers common challenges in the medical software industry and how requirements management solutions can help you.
Medical device companies face different and greater challenges than non-medical device companies. Regulatory hurdles, the stakes for end-users, and the complexities of project management pose significant roadblocks. Any software that helps you navigate these challenges also becomes a healthcare risk management software. Here are some common challenges companies face in medical device software development:
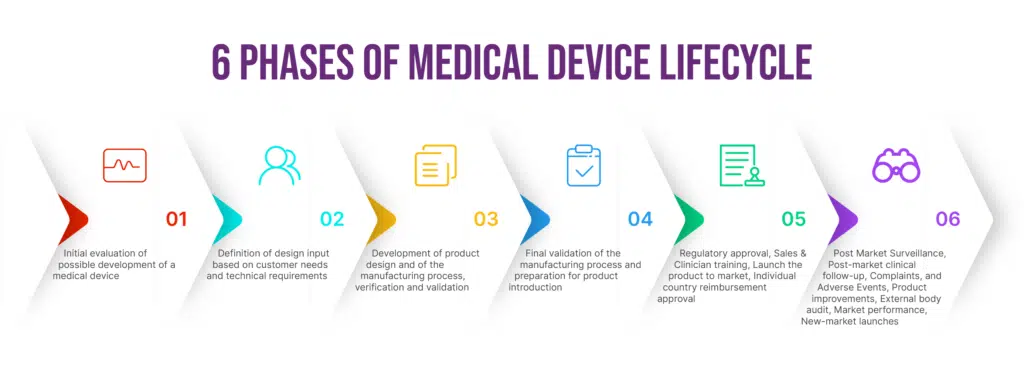
Medical device software is a critical component in modern healthcare solutions. Its development demands a meticulous approach to ensure functionality, safety, and regulatory compliance. Requirements Management tools serve as the bedrock for success, providing a structured framework to capture, analyze, and trace every aspect of software requirements.
The traditional method of requirements management in healthcare development is unsuited for medical device software development. With this method, hardware and software teams would meet these needs separately after agreeing on stakeholder needs. But they used different tools and methodologies, leading to traceability issues and expensive rework.
This leads to a long process of finding and fixing problems between the two teams and can take up a lot of time that could have been used to make the device better.
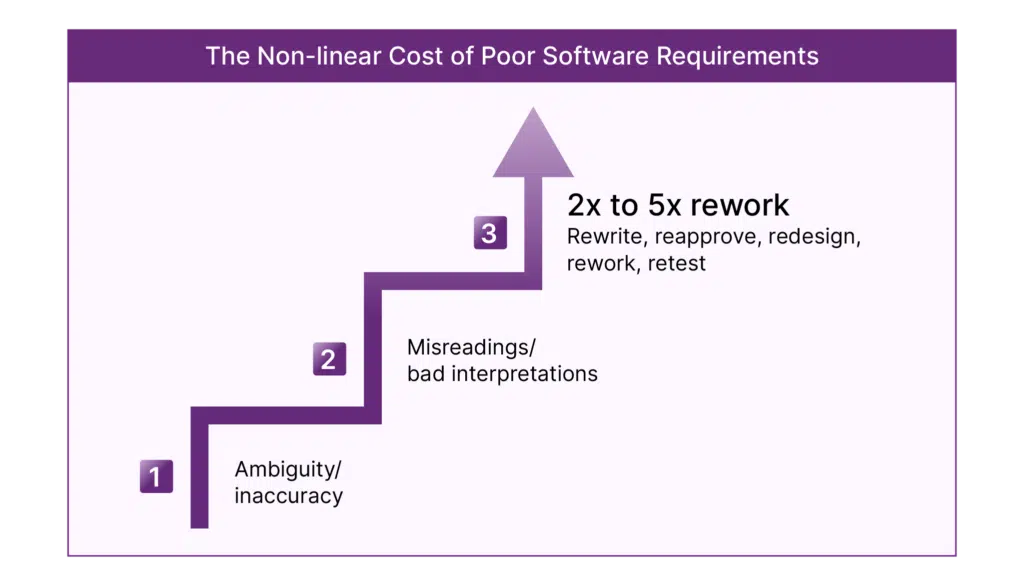
To manage product complexity, user interfaces, regulations, and patient data, requirements management tools are necessary. They streamline development and deployment of software and mitigate risks during the development of medical devices and their software. The best requirements management solutions leverage widely used existing platforms like Azure DevOps and turn it into a single source of truth. They also manage healthcare risk management by extension.
In healthcare, meeting regulatory needs is key to medical device success. It is estimated that the global market has two million medical devices in total, divided into approximately 7000 device groups. These devices must meet global, local, and category specific regulatory standards.

Regulatory bodies like the Food and Drug Administration (FDA) in the United States and the European Medicines Agency (EMA) in Europe set these standards. In the US alone, the FDA requires medical devices to satisfy at least seven basic regulatory requirements. These standards cover a wide range of medical device aspects, from design to post-launch surveillance. An ordinary blood glucose monitor may have to adhere with the following (non-exhaustive) list of standards:
| Function | Regulation (if relevant) | Compliance Standard | Level |
| Infection Control | CDC guidelines on infection control during blood glucose monitoring | CLIA Certificate of Compliance (CoC) or Certificate of Accreditation (CoA) | National (US) |
| Device Design and Use | FDA guidelines for self-monitoring blood glucose test systems | ISO 15197 | National (US) |
| E-Signatures | Title 21 of the Code of Federal Regulations, established by the FDA. | CFR Part 11 | National (US) |
| Risk Management | N/A | ISO 149715 | International |
| Software Lifecycle Processes | N/A | IEC 623046 | International |
| Health Software Safety | N/A | IEC 82304-17 | International |
| Medical Electrical Equipment Safety | N/A | IEC 60601-18 | International |
Requirements management tools play a crucial role in aligning medical products with standards. A team must document, analyze, trace, and prioritize requirements they must satisfy. By effectively managing requirements, medical device companies can ensure that their products not only meet regulatory standards, but also deliver safe and effective solutions to healthcare providers and patients.
Precision is non-negotiable in the medical field, where the stakes are high, and accuracy is paramount. Requirements Management tools enable development teams to articulate and document software requirements with meticulous detail.
When creating medical device software, teams may use multiple features that requirements management tools provide, like:
A practical example of a requirements elicitation technique is interviewing stakeholders. The most important factor in elicitation through interviews are the questions you ask. Elicitation is best done early in the prototype design. A pre-existing and editable batch of FAQs can help you gather the right questions for eliciting. An added AI tool can also generate project-specific questions by ingesting raw requirements data.
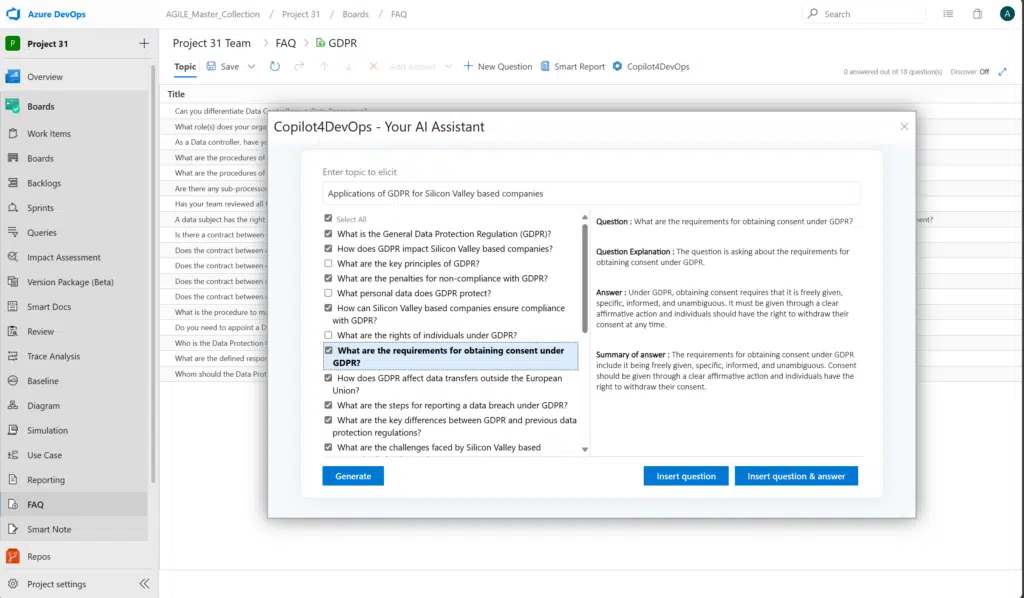
Many errors in documentation may creep in between tight deadlines and high-stakes competition. More than a third of software engineers consider poor documentation one of their biggest challenges. This is more critical for medical device software development.
Easily creating structured Word-like requirements documents within an ALM like Azure DevOps can help reduce these errors.
The complexity of harmonizing hardware and software development can create errors. It’s easy to lose track of how tasks are related. Creating traceability matrices to trace requirements from end-to-end can help reduce these errors. It also helps project efficiency, component testing, impact analysis, and team communication.
In such cases, it is helpful to output reports of anything and everything from your project in one click.
It’s even better if you can output documents as PDF, Word, or HTML.
Modern Requirements4DevOps is a solution designed to streamline the requirements management process in medical device software development. Its key features include traceability, collaboration, and change management, which enhance project efficiency and ensure alignment with regulatory standards. It also has a groundbreaking AI tool called Copilot4DevOps, which increases requirements quality through elicitation, analysis, and a lot more.
By leveraging Modern Requirements4DevOps, healthcare organizations like Bayer and Red Cross are ensuring better regulatory compliance, project efficiency, and reduced development costs. Here’s how you can use it too.
Effective requirements management is crucial in medical device software development. Though requirements management tools are the best way to level-up your project, there are also some best practices that should apply regardless of the tools you use:
As the medical device market grows, the difficulty of balancing hardware, software, and regulation will increase. A robust requirements management tool is your ally when managing these complexities.
Newly by artificial intelligence, these tools streamline development, ensure regulatory alignment, and reduce risks. They can do this for both medical device software and hardware. As the sector evolves, these tools will be pivotal for success, driving innovation, and leading to better patient outcomes.
✅ Define, manage, and trace requirements within Azure DevOps
✅ Collaborate seamlessly across regulated teams
✅ Get started for FREE—no credit card required
Join Modern Requirements at Info-Tech Live 2026 and witness firsthand...
Learn how to automate FedRAMP SSP and POA&M documentation to...
Learn how to trace, maintain, prioritize, and control requirements throughout...
End-to-end requirements management in Azure DevOps.
AI-powered assistance for DevOps workflows.
Autonomous AI agents for DevOps execution.
Real-time data sync across tools and systems.
Designed to work natively within Azure DevOps, Modern Requirements extends the platform with powerful capabilities that help teams capture, manage, and validate requirements more effectively.